Every year, stroke strikes the world like a silent emergency hiding in plain sight. Ischemic stroke — caused by a blood clot blocking blood flow to the brain — is the second leading cause of death on the planet, and the single largest cause of long-term disability. The global cost exceeds 890 billion US dollars annually. And yet the most devastating fact is not the number of people it kills. It is the number of people it reaches before medicine can.
The brain does not wait. Every minute a stroke goes untreated, approximately two million neurons die. The current gold-standard treatments — intravenous clot-busting drugs and mechanical clot removal — are both hospital procedures. They require imaging equipment, specialist teams, and a time window so narrow that more than 85 percent of stroke patients worldwide never receive them at all. By the time the ambulance arrives, by the time the scan is complete, by the time the team assembles — for most people, the damage is already done.
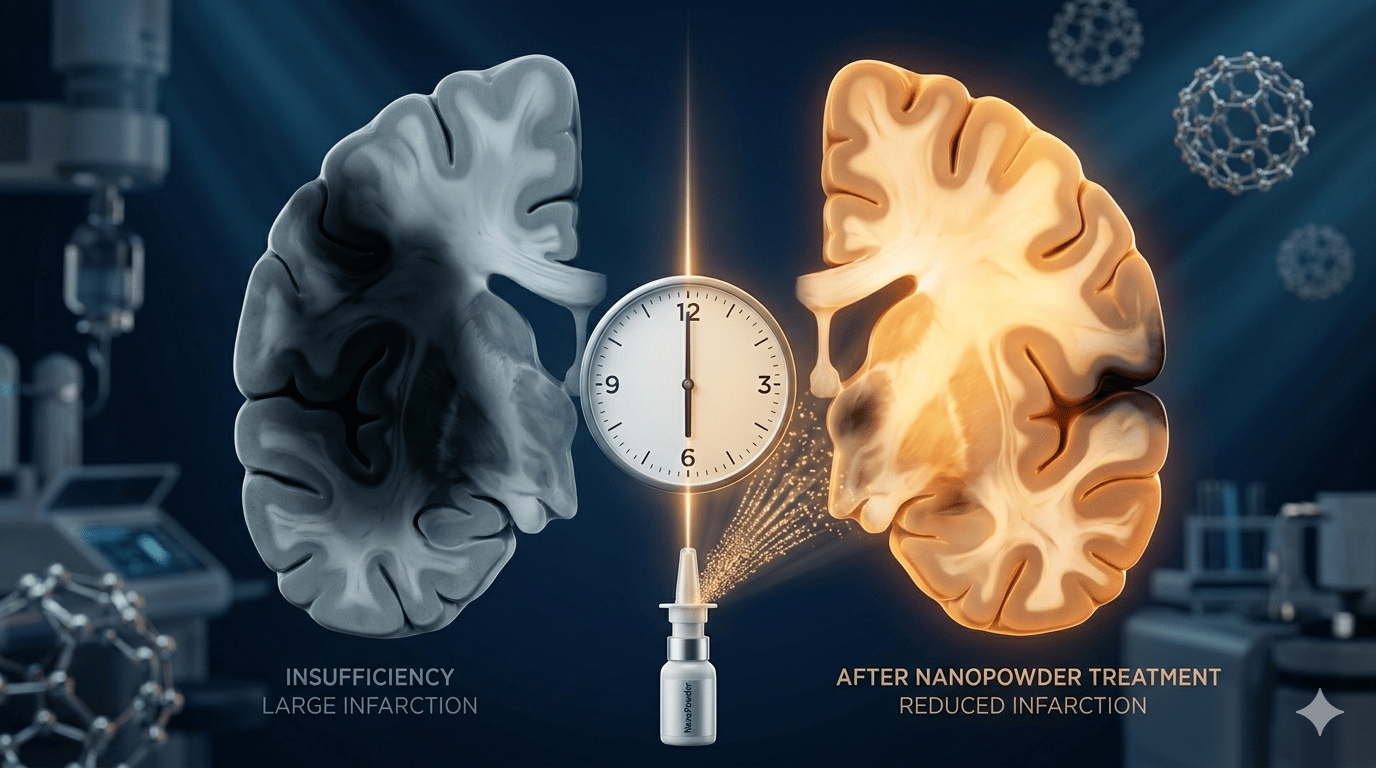
That is the wall. And it is the wall that a team at the University of Hong Kong's Li Ka Shing Faculty of Medicine has spent more than a decade trying to break through.
This is an AMAZING moment because what they have built is not a hospital treatment. It is a nasal spray — portable, needle-free, and designed to be used in the back of an ambulance, in a care home, or eventually, in any household in the world.
The technology is called NanoPowder. It is built on a platform the researchers call "Nano-in-Micron" — a system that packages neuroprotective drugs inside nanoparticles, then compresses those nanoparticles into a micron-sized powder fine enough to inhale. When the powder enters the nasal cavity, it deposits precisely in the target area and, on contact with nasal mucus, rapidly breaks apart back into nanoparticles. Those nanoparticles then travel the nose-to-brain pathway — a direct biological route that bypasses the blood-brain barrier entirely.
The blood-brain barrier is the reason 90 percent of central nervous system drugs fail in clinical trials. The brain protects itself from foreign substances with extraordinary efficiency. The NanoPowder spray does not try to force through it. It goes around it.
In preclinical animal studies, using the spray within 30 minutes of stroke onset reduced ischemic brain infarction — the volume of dead and dying brain tissue — by more than 80 percent, while effectively protecting both motor and neurological function. The drug has shown potential to reduce brain inflammation, prevent cell death, and protect the blood-brain barrier itself. Critically, in tests where the spray was mistakenly administered during hemorrhagic stroke — a different and rarer type — no significant toxicity or side effects were observed.
Professor Aviva Chow Shing-fung, who co-leads the research, put the mission plainly: the spray allows patients to receive initial protection before they reach hospital, significantly slowing the death of brain cells under ischemic conditions and preserving still-viable brain tissue — buying time for the treatments that follow.
Why does this matter to you? Because stroke does not announce itself with paperwork. It arrives in the middle of the night, at a family dinner table, during a morning walk. The people most likely to be present are not doctors. They are partners, adult children, care workers, and bystanders. A technology that gives those people something to do — something that works — in the minutes before professional help arrives is not merely a medical advance. It is a shift in who gets to survive.
The research team's roadmap places clinical trials at 2030, with rollout beginning through paramedics and emergency services, then expanding to care homes and high-risk residential settings, and ultimately to retail pharmacies as an over-the-counter emergency tool. The spray's compact size and portability make it well suited to all three environments.
The NanoPowder's underlying platform also has broader potential applications — including Alzheimer's disease, motor neuron disease, and meningitis — conditions that share the same fundamental obstacle of drug delivery across the blood-brain barrier.
I want to be honest with you about what this is not. NanoPowder has not yet been tested in human clinical trials. Animal models are a critical and necessary step, but they do not guarantee the same outcomes in human patients. The road from preclinical success to pharmacy shelf is long, carefully regulated, and genuinely uncertain. Clinical trials are not expected until 2030. What we have today is a world-first proof of concept with striking early data — and a research team with a decade of serious, awarded work behind them.
The history of medicine is full of breakthroughs that were announced too early and too loudly. This is not that. This is a measured, peer-reviewed, internationally recognized finding that deserves serious attention — and honest hope.
Stroke has always been a race between biology and medicine. For most of human history, biology has been winning — because medicine could not get to the brain fast enough. If NanoPowder fulfills its preclinical promise in human trials, that race changes. The starting line moves from the emergency room to the moment the spray is administered. And for the millions of people who never make it to the emergency room in time, that difference is everything.
Sources:
HKUMed official press release, "HKUMed develops world-first nasal spray as prehospital emergency aid for ischemic stroke," May 5, 2026: https://www.hku.hk/press/news_detail_29102.html
South China Morning Post, "Hong Kong researchers develop 'world-first' nasal spray for rapid stroke aid," May 2026: https://www.scmp.com/news/hong-kong/health-environment/article/3352496/hong-kong-researchers-develop-world-first-nasal-spray-rapid-stroke-aid
BioSpectrum Asia, "Hong Kong develops world-first nasal spray as prehospital emergency aid for ischemic stroke," May 2026: https://www.biospectrumasia.com/news/30/27598/hong-kong-develops-world-first-nasal-spray-as-prehospital-emergency-aid-for-ischemic-stroke.html
Support the Mission
Fuel the Research: Love these daily "Yeah!" moments? [Buy Henry P. a Coffee]
Start Your Own Revolution: This newsletter is built on [Beehiiv Partner Link]. Start your own for free today!
